Introduction
Gabapentin, a drug originally prescribed for the treatment of seizures, is now frequently used off-label for pain management. However, its use can cause side effects such as drowsiness and dizziness, which may impair driving and pose significant safety risks. Considering these issues, the National Safety Council’s Alcohol, Drugs, and Impairment Division (NSC-ADID) has recently upgraded gabapentin’s classification from a Tier II to a Tier I substance, adding it to the standard routine drug testing panel. For this purpose, NSC-ADID established a cutoff concentration of 50 ng/mL in oral fluid.
Our goal for this application note is to use a simple sample preparation method and a fast analysis technique for gabapentin analysis in oral fluid (OF) sample.
The LDTD-MS/MS system using Axino Ion Source offers specificity combined with an ultra-fast analysis for an unrivaled analysis method. To develop this application, we focused on performing a simple sample preparation.
Sample Preparation Method
Sample Collection
Oral fluid sample collection is performed with the Quantisal device, which is FDA 510(k) cleared for the collection of oral fluid for drug analysis.
Automated Sample Extraction (SALLE)
The calibration curve was prepared in negative oral fluid samples.
- In a 12 X 75 mm borosilicate tubes, 10 µL internal standard solution (gabapentin-d10 at 25 µg/mL in acetonitrile) are added.
- 125 µL of spiked negative OF in Quantisal buffer (1:3) are added.
- Vortex (1100 rpm/30 seconds)
- 500 µL of extraction solution are added.
- 1000 µL of dilution solution are added.
- Vortex (1100 rpm/60 seconds)
- Centrifuge (5000 rpm/5 min.)
- Transfer 50 µL of the aqueous phase (upper layer) in a 0.5 mL microcentrifugation tube.
- In the 0.5 mL microcentrifugation tube, add 150 µL of desorption solution.
- Vortex (1100 rpm/10 secoonds)
- Spot 5 µL of upper layer on a Domino plate
- Dry 8 minutes at 40°C
LDTD®-MS/MS Parameters
LDTD
Model: Axino, Phytronix
Carrier gas: High
Laser pattern: 6-second ramp to 65% power
MS/MS
MS model: Q-Trap System® 5500, Sciex
Ionization: APCI
Analysis Method: Positive MRM mode
CUR: 20
CAD: 8
| Drugs | Transition | CE |
|---|---|---|
| Gabapentin | 172.0 → 137.0 | 25 |
| Gabapentin-d10 | 182.0 → 147.0 | 25 |
Results and Discussion
Validation Test
To evaluate method precision and accuracy, a three-point calibration curve was employed (1 x cutoff, 2 x cutoff, and 5 x cutoff). For the screening approach, a single-point calibration at the cutoff level was used, applying a linear regression constrained to pass through the origin. The peak area against the internal standard (IS) ratio was used to normalize the signal.
Linearity
For inter-run results, linearity was evaluated. Correlation coefficient (r) must be higher than 0.97. Linearity obtained was higher than 0.991.
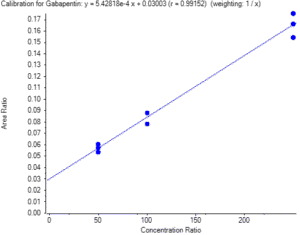
Figure 1 – Three-point calibration curve for Gabapentin.
Inter-run Precision and Accuracy
Spiked samples around the decision point (Cal-1X, two times cut-off: Cal-2X, and five times cut-off: Cal-5X) and blank solutions are used to validate the precision of the method. For quality control samples, these samples are spiked in real negative matrices. For QC-0.5X, 50% cut-off are spike in sample, and for QC-2X, 200% cut-off are spiked in sample. Replicate extractions are deposited on a Domino LazWell™ plate and dried before analysis.
- Each concentration must not exceed 20% CV for 66.6% of samples
- Each concentration must not exceed 20%Bias for 66.6% of samples.
For the inter-run precision experiment, each fortified sample set is analyzed in triplicate. Table 2 shows the inter-run precision and accuracy results for gabapentin. %CV and %Bias were below 20%.
| Gabapentin | Cal-1X | Cal-2X | Cal-5X |
|---|---|---|---|
| Conc (ng/mL) | 50 | 100 | 250 |
| N | 6 | 6 | 6 |
| Mean (ng/mL) | 49.2 | 102.0 | 248.7 |
| %CV | 9.7 | 7.5 | 5.1 |
| %Bias | 1.5 | -2.0 | 0.5 |
Run Acceptance Criteria for Intra-run
Intra-run validation tests were carried out using a one-point calibration curve and linear through zero regression. Cutoff and Qcs were analyzed in triplicate. Table 3 shows intra-run results.
- Blank samples must be detected as negative.
- At Cal-1X concentration, %CV must not exceed 20% for at least 66.7% of this standard.
- At QC-0.5X, at least 66.7% of samples must be detected as negative.
- At QC-2X, 66.7% of samples must be detected as positive.
| Gabapentin | Run 1 | Run 2 |
|---|---|---|
| Blank | Negative | Negative |
| Conc cutoff (ng/mL) | 50 | 50 |
| N | 3 | 3 |
| Mean (ng/mL) | 48.8 | 50.0 |
| %CV | 6.7 | 6.4 |
| QC-0.5X | Negative (3/3) | Negative (3/3) |
| QC-2X | Positive (3/3) | Positive (3/3) |
Multi-Matrix Study
Real OF samples (N=4) have been tested with this method. These samples were readily known to be exempt from gabapentin. All samples were determined to be negative by Axino-MS/MS analysis. Results are reported in Table 4
| Axino | Real | |
|---|---|---|
| M1 | Negative | Negative |
| M2 | Negative | Negative |
| M3 | Negative | Negative |
| M4 | Negative | Negative |
Conclusion
Axino Ion Source® combined to a Sciex Q-Trap 5500 mass spectrometer system allows ultra-fast (10 seconds per sample) for screening gabapentin in oral fluid using a simple and efficient sample preparation method.