Introduction
Glipizide, a second-generation of sulfonylurea, is effective in controlling the blood glucose in patients with noninsulin-dependent diabetes mellitus1. During antidiabetic therapy, it is critical to monitor the plasma concentration of glipizide.
Our goal for this application note is to use an automated sample preparation method for the quantification of Glipizide in plasma using a single operation in LDTD-MS/MS.
LDTD-MS/MS offers specificity combined with an ultra-fast analysis for an unrivaled quantification method. To develop this application, we focused on performing a quick and simple sample preparation. Glipizide is analyzed, and results are obtained in less than 5 seconds per sample.
Luxon Ionization Source
The Luxon Ion Source® (Figure 1) is the second-generation sample introduction and ionization source based on the LDTD® technology for mass spectrometry. Luxon Ion Source® uses Fiber-Coupled Laser Diode (Figure 2) to obtain unmatchable thermal uniformity providing more precision, accuracy, and speed. The process begins with dry samples which are rapidly evaporated using indirect heat. The thermally desorbed neutral molecules are carried into a corona discharge region. High efficiency protonation and strong resistance to ionic saturation characterize this type of ionization and is the result of the absence of solvent and mobile phase. This thermal desorption process yields high-intensity molecular ion signal in less than 1 second sample-to-sample and allows working with very small volumes.
Figure 1 – Luxon Ion Source®
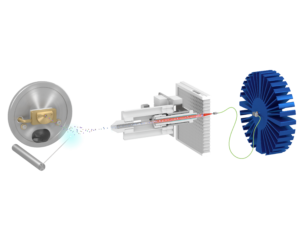
Figure 2 – Schematic of the Luxon Ionization Source
Sample Preparation Method
Automated Sample Extraction
Glipizide, stock solutions were prepared in methanol. EDTA-K2 plasma was used as a negative matrix then spiked to generate a calibration curve and QCs.
Plasma samples were transferred into barcoded tubes, readable by the Azeo extraction system. Each barcoded vial was scanned by the Azeo Liquid Handler and an automatic batch file was created.
The Azeo Liquid Handler (Figure 3) is used to extract the samples using the following conditions:
- 10 µL of Internal standard (Glipizide-d11 at 1000 ng/mL in Acetonitrile:Water (1:1)) were added to a deep-well plate placed on the Lumo Vortexer.
- 50 µL of plasma sample were transferred from the vials to a deep-well plate placed on the Lumo Vortexer.
- Mix (30 seconds at 1000 rpm).
- 50 µL of extraction buffer were added into a deep-well plate.
- Mix (30 seconds at 1000 rpm).
- 300 µL of Methyl-Tert-Butyl Ether (MTBE) were added into a deep-well plate.
- Mix (30 seconds at 1000 rpm).
- Centrifugation at 5000 rpm for 2 minutes for phase separation.
- Spot 7 µL upper layer phase onto a coated LazWell™ 96 plate.
- Dry at room temperature.

Figure 3 – Automated extraction system.
LDTD®-MS/MS Parameters
LDTD
Model: Luxon SH-960, Phytronix
Carrier gas: 3 L/min (air)
Laser pattern:
- 3-second ramp to 65% power
MS/MS
MS model: LCMS-8060, Shimadzu
Scan Time: 50 msec.
Total run time: 5 seconds per sample
Ionization: APCI
Analysis Method: Positive MRM mode
Table 1 – MRM transitions for LDTD-MS/MS
| Transition | CE | |
| Glipizide | 446.2 → 321.1 | 20 |
| Glipizide-d11 | 457.2 → 321.1 | 20 |
Results and Discussion
Data preparation process
Mass spectrometers are data acquisition systems that were not designed to deal with signals of a few seconds per sample. The synchronization sequence adds 6 to more than 15 seconds between each sample. To bypass this delay, all samples are acquired in a single file (Figure 4). To allow the analysis of such data, Cascade software is designed to detect, split, and integrate every sample peak acquired in a single file.
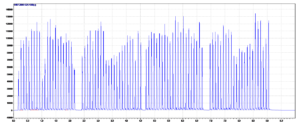
Figure 4 – Single file mass spectrometer data for 96 samples. Glipizide-d11 transition.
Validation Test
Calibration curves ranging from 10 to 1000 ng/mL and QCs were prepared in EDTA-K2 plasma. Replicate extractions were deposited onto a LazWell™ plate and dried before analysis. The peak area against the internal standard (IS) ratio was used to normalize the signal.
Linearity
The calibration curves were plotted using the peak area ratio and the nominal concentration of standards. For the linearity test, the following acceptance criteria was used:
- Linear regression (r2) must be ≥ 99
Figure 5 shows a typical calibration curve result for Glipizide. Table 2 shows the coefficients of determination (R2) of six different runs.
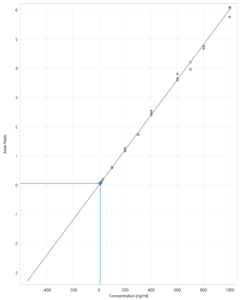
Figure 5 – Glipizide calibration curve.
Table 2 – Linearity results
| R2 | |
| Run 1 | 0.99912 |
| Run 2 | 0.99925 |
| Run 3 | 0.99902 |
| Run 4 | 0.99970 |
| Run 5 | 0.99913 |
| Run 6 | 0.99908 |
Precision and Accuracy
For the accuracy and precision evaluation, the following acceptance criteria were used:
- Each concentration must not exceed 20% CV.
- Each concentration must be within 100 ± 20% of the nominal concentration.
For the inter-run precision and accuracy experiment, each QCs were analyzed in triplicate in six different runs. For the intra-run precision and accuracy experiment LLOQ, QC-L, QC-M, QC-H and ULQC were analyzed in six replicates. Table 3 and 4 show the intra and inter-run precision and accuracy results for Glipizide. The obtained %CV was below 15% and the accuracy was within 15% of the nominal value.
Table 3 – Intra-Run Precision and Accuracy
| Glipizide | LLOQ | QC-L | QC-M | QC-H | ULQC |
| Conc (ng/mL) | 10 | 30 | 300 | 700 | 1000 |
| N | 6 | 6 | 6 | 6 | 6 |
| Mean (ng/mL) | 10.0 | 29.1 | 300.5 | 718.8 | 1001.4 |
| %CV | 3.6 | 6.5 | 1.5 | 1.5 | 1.5 |
| %Nom | 99.7 | 96.8 | 100.2 | 102.7 | 100.1 |
Table 4 – Inter-Run Precision and Accuracy
| Glipizide | LLOQ | QC-L | QC-M | QC-H | ULQC |
| Conc (ng/mL) | 10 | 30 | 300 | 700 | 1000 |
| N | 18 | 18 | 18 | 18 | 18 |
| Mean (ng/mL) | 9.5 | 32.6 | 302.8 | 699.4 | 993.2 |
| %CV | 6.4 | 6.5 | 5.5 | 4.6 | 2.9 |
| %Nom | 94.8 | 108.7 | 100.9 | 99.9 | 99.3 |
Stability
Wet stability: Following the extraction, the extracted sample was kept at 4°C in closed containers. After 1 day, extracted samples are spotted on a LazWell™ plate, dried and analyzed. Precision and accuracy are reported in Table 5. All the results are within the acceptable criteria range for 1 day at room temperature.
Dry stability: Extracted samples are spotted onto a LazWell™ plate, dried and kept at room temperature for 1 hour before analysis. The precision and accuracy results are reported in Table 5. All the results are within the acceptable criteria range for 1 hour at room temperature.
Table 5 – Wet and Dry Stability of Glipizide
| Glipizide | Wet stability (1 day/4°C) | Dry stability (1 hour / RT) | ||||
| QC-L | QC-M | QC-H | QC-L | QC-M | QC-H | |
| Conc. (ng/mL) | 30 | 300 | 700 | 30 | 300 | 700 |
| N | 3 | 3 | 3 | 3 | 3 | 3 |
| Mean (ng/mL) | 34.8 | 314.5 | 717.0 | 32.4 | 283.4 | 671.0 |
| %CV | 1.2 | 1.9 | 0.2 | 9.3 | 3.7 | 1.8 |
| %Nom | 115.8 | 104.8 | 102.4 | 108.1 | 94.5 | 95.9 |
Matrix effect study
Six different plasma samples were spiked at QC-M level, extracted, and analyzed. The precision and accuracy results of the different matrix spiked are reported in Table 6. All the results are within the acceptable criteria range.
Table 6 – Matrix effect result of Glipizide
| Glipizide | ||||||
| M1 | M2 | M3 | M4 | M5 | M6 | |
| Conc. (ng/mL) | 300 | 300 | 300 | 300 | 300 | 300 |
| N | 3 | 3 | 3 | 3 | 3 | 3 |
| Mean (ng/mL) | 341.3 | 297.3 | 313.5 | 339.5 | 324.4 | 335.8 |
| %CV | 3.1 | 3.8 | 5.2 | 1.1 | 0.6 | 0.5 |
| %Nom | 113.8 | 99.1 | 104.5 | 113.2 | 108.1 | 111.9 |
Conclusion
The Luxon Ion Source® combined with Shimadzu LCMS-8060 mass spectrometer system allows ultra-fast (5 seconds per sample) analysis of Glipizide in plasma.
Reference
- Brogden et Al. (1979). Drugs 18:329-353